 
In the laboratory, for example, the masses of compounds and elements used by chemists typically range from milligrams to grams, while in industry, chemicals are bought and sold in kilograms and tons. Because the masses of individual atoms are so minuscule (on the order of 10 −23 g/atom), chemists do not measure the mass of individual atoms or molecules. The problem for Dalton and other early chemists was to discover the quantitative relationship between the number of atoms in a chemical substance and its mass. We also described the law of multiple proportions, which states that the ratios of the masses of elements that form a series of compounds are small whole numbers. In Dalton’s theory each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. The same calculation can also be done in a tabular format, which is especially helpful for more complex molecules: Indeed, hydrogen has been a key element in establishing quite reasonable theories of acidity and basicity, which came to be viewed as proton transfer reactions in the Brønsted-Lowry theory.\right ) \right ] \) On the contrary, the H − anion is a very strong base and a strong reducing agent, whereas H is a voracious acid and a powerful oxidizer non (or very slightly)-hydrated protons present in a superacidic environment readily convert alkanes into carbocations 8. It was only in 1984 that Kubas described the coordination of molecular H 2 to transition metals 7. 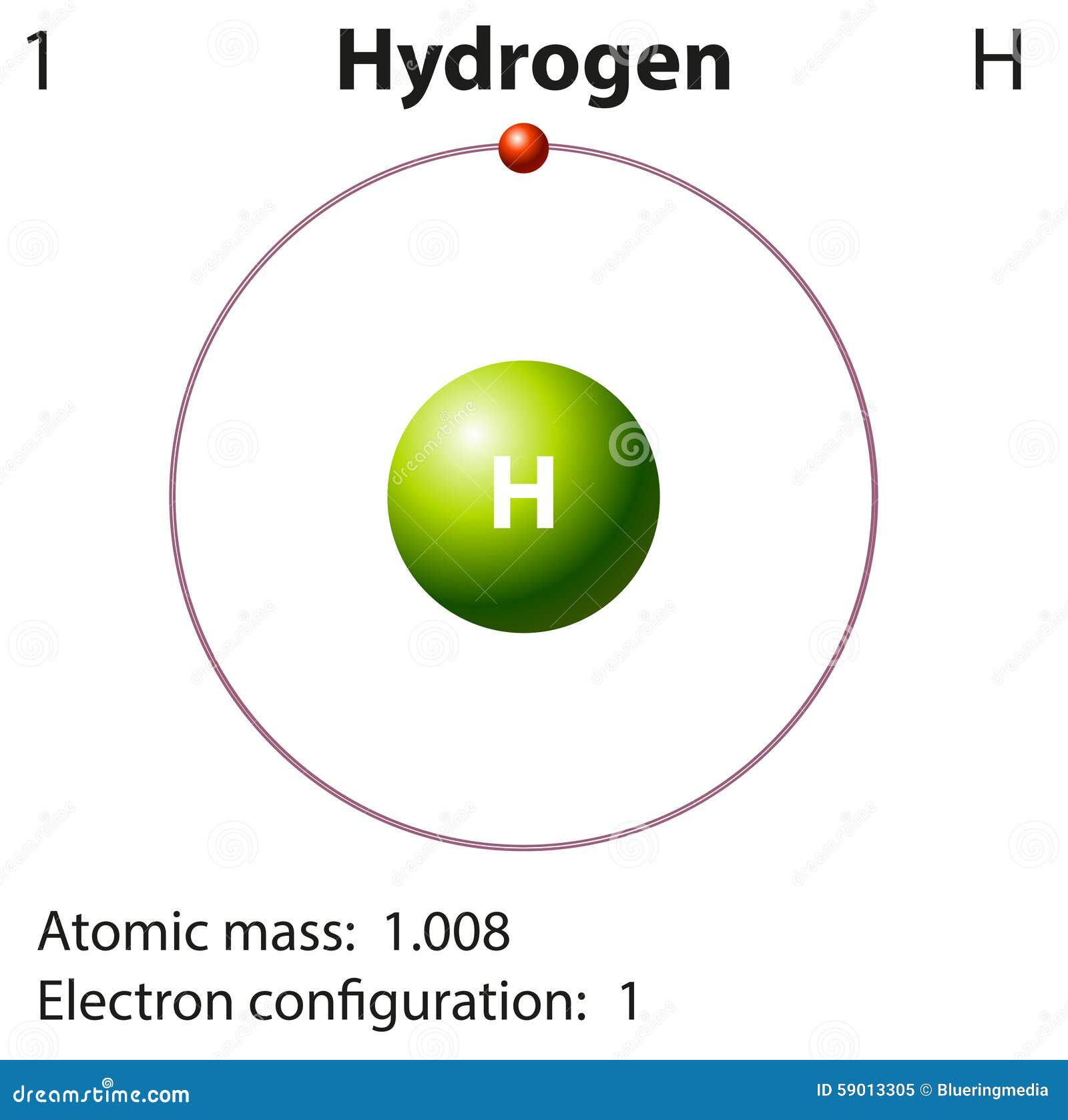
The H 2 molecule - isoelectronic to the closed-shell He atom in the unified atom model - is quite inert. The oxidation states of hydrogen span from −1 (hydride), through 0 (elemental), to 1 (proton), with very different physicochemical properties for each species. These two species have served as test beds for rigorous critical evaluations of diverse quantum mechanical models and approximations 6. Molecular H 2 filled one of the first balloons used to carry people in 1783 (pictured), and the fuel tanks of rockets two centuries later, permitting the inquisitive to explore further and further.īecause H and H 2 constitute the prototypical atom and molecule, respectively, they have been extensively used by theoreticians for over a century - since the birth of quantum mechanics. The evolved H 2 gas is an excellent, ultra-light energy carrier, and very promising as a fuel - abundant and environmentally friendly as its oxidation produces water. It is this process that we try to achieve today, although with a much smaller electric bill, through a photochemical process 2. Conversely, in 1800 Nicholson and Carlisle (shortly followed by Ritter) managed to decompose water into its elemental constituents using electrolysis. This discovery inspired Lavoisier to call the substance 'hydro-gen', meaning water-former, in 1783. In 1671 Robert Boyle described the formation of gas bubbles from the reaction of iron filings with acid, but it was Cavendish who recognized H 2 (which he referred to as 'inflammable air') as a substance distinct from other gases, which, when it was burnt in 'dephlogisticated air' (oxygen) produced water. Van Helmont was the first to find out that although hydrogen was combustible in air, it could not support combustion by itself. 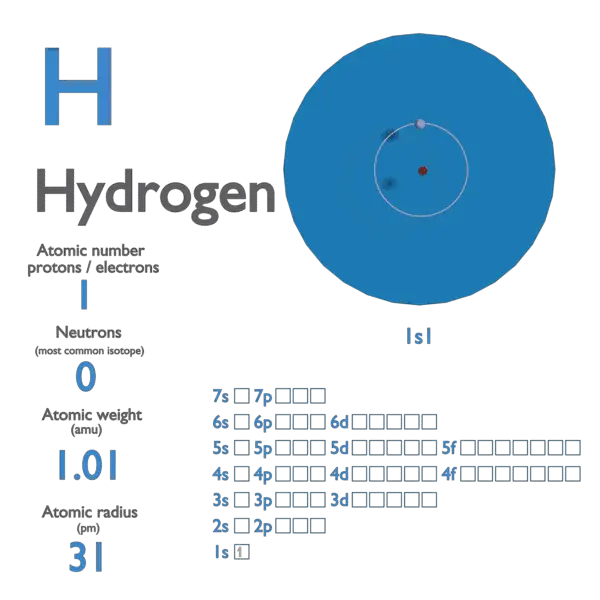
Credit: LIBRARY OF CONGRESS / SCIENCE PHOTO LIBRARY 
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
